The recurrence of head and neck squamous cell carcinoma (HNSCC) after surgical resection continues to pose a major challenge to cancer treatment. Advanced HNSCC exhibits a low response rate to immune checkpoint blockade (ICB), while photothermal therapy (PTT) can increase the infiltration of immune cells to make tumors more susceptible to cancer immunotherapy.
In this regard, Professor Guang Yang’s 3Bio Group at College of Life Science and Technology, Huazhong University of Science and Technology,Professor Zhi-Jun Sun at school & hospital of stomatology ,Wuhan University reported a novel multifunctional nanocomposite comprised of oxidized bacterial cellulose (OBC), thrombin (TB), and gold nanocages (AuNCs) containing anti-programmed death 1 (PD-1) antibody as the title of Implantable versatile oxidized bacterial cellulose membrane for postoperative HNSCC treatment via photothermal-boosted immunotherapy in the international journal of NANO RESEARCH.

The newly developed TB/aPD-1@AuNCs/OBC composites demonstrated a remarkable ability toward facilitating the relief of the immunosuppressive TME and inducing systemic immune responses. The aPD-1 released from this system allowed the combination with local ICB therapy to inhibit local recurrence of the residual tumor.Specifically, aPD-1@AuNC under NIR irradiation could generate ROS to induce pyroptosis and release intracellular contents (i.e., HMGB1, LDH, and ATP), which triggered T-cellmediated robust tumor eradication due to the enhanced DCs maturation process and presentation of tumor-specific antigens, which enabled the PTT therapy boosted the immunotherapy mediated by aPD-1 and resulted in excellent antitumor efficacy in SCC7-tumor-bearing mice. Additionally, the Food and Drug Administration (FDA) has approved BC products in the biomedical field recently, which have demonstrated the feasibility and safety of TB/aPD-1@AuNCs/OB. The TB can be mixed within the tumor resection blood after surgery to make the TB/aPD-1@AuNCs/OBto adhere postoperative sites, which is more in line with the operation habits of surgeon. Therefore, the versatile OBC-based platform could be adapted in “real-world practice" and might have promising clinical applications for reducing the tumor recurrence in postoperative HNSCC tumor treatment.
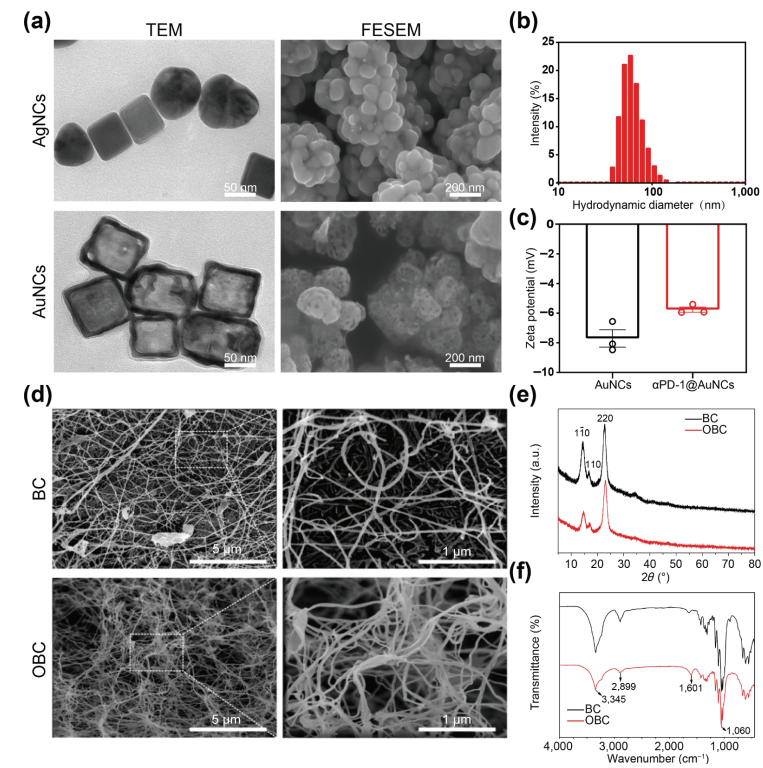
Figure 1. Physicochemical characterizations of AgNCs, AuNCs, BC, and OBC.(a) Representative TEM and FESEM images of AgNCs and AuNCs.(b) Intensity distribution histogram of AuNCs determined using DIS.(c) Zeta potential of AuNCs and aPD-1@AuNCs. (d) Typical FESEM images of BC and OBC. (e) XRD patterns of BC and OBC. (f) FTIR spectra of BC and OBC.

Figure.2. Physicochemical properties and biodegradability characterization of OBC-based implants.(a) Representative FESEM images of OBC, TB/OBC,aPD-1@AuNCs/OBC,and TB/aPD-1@AuNCs/OBC. (b) Representative images of BC, OBC,TB/OBC,and TB/aPD-1@AuNCs/OBC. (c) XRD patterns of OBC, TB/OBC,aPD-1@AuNCs/OBC, and TB/aPD-1@AuNCs/OBC.(d) TGA curves of OBC, TB/OBC, aPD-1@AuNCs,aPD-1@AuNCs/OBC,and TB/aPD-1@AuNCs/OBC.(e) H&E staining of BC and OBC in the tissue after implantation into the surgical sites of SCC7 tumor-bearing mice for 14 days.
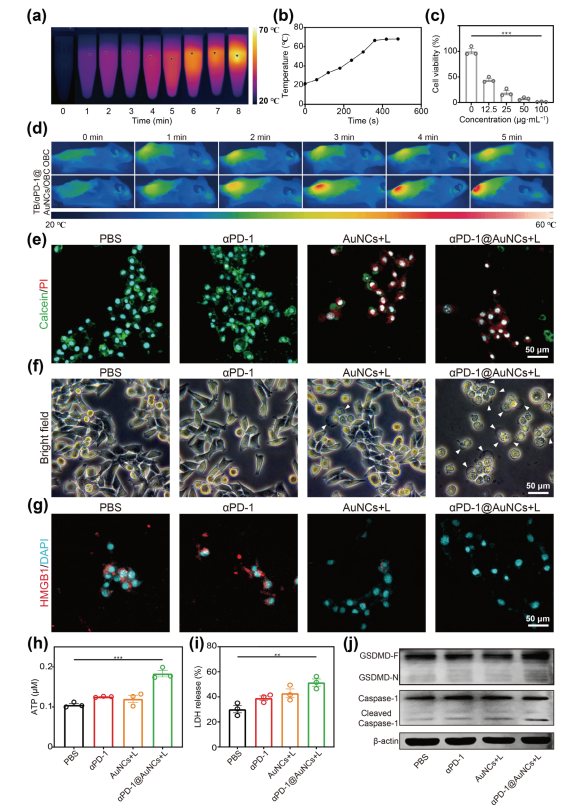
Figure.3. aPD-1@AuNCs induce tumor cell death in vitro. (a) Photothermal infrared images of aPD-1@AuNCs.(b) The temperature variation curve of aPD-1@AuNCs. (c) Cytotoxicity of aPD-1@AuNCs at different concentrations.(d) Photothermal infrared images of SCC7 tumor-bearing mice treated with PBS under NIR irradiation or TB/aPD-1@AuNCs/OBC under NIR irradiation. (e) Microscopic images of Calcein-AM/PI-stained SCC7 cells after different treatments. (f) Optical microscopy images of SCC7 cells treated with PBS, aPD-1, AuNCs+L, and aPD-1@AuNCs+L.(g) Microscopic images of HMGB1/DAPI-stained SCC7 cells treated with PBS, aPD-1, AuNCs+L, and aPD-1@AuNCs+L. (h) ATP secretion from SCC7 cells treated with PBS, aPD-1, AuNCs+L, and aPD-1@AuNCs+L.(i) LDH release in the supernatant was assessed in SCC7 cells treated with PBS, aPD-1, AuNCs+L, and aPD-1@AuNCs+L.(i)Expression levels of pyroptosis-associated proteins inSCC7 cells (GSDMD-F, GSDMD-N, and cleaved Caspase 1) were analyzed by western blot.
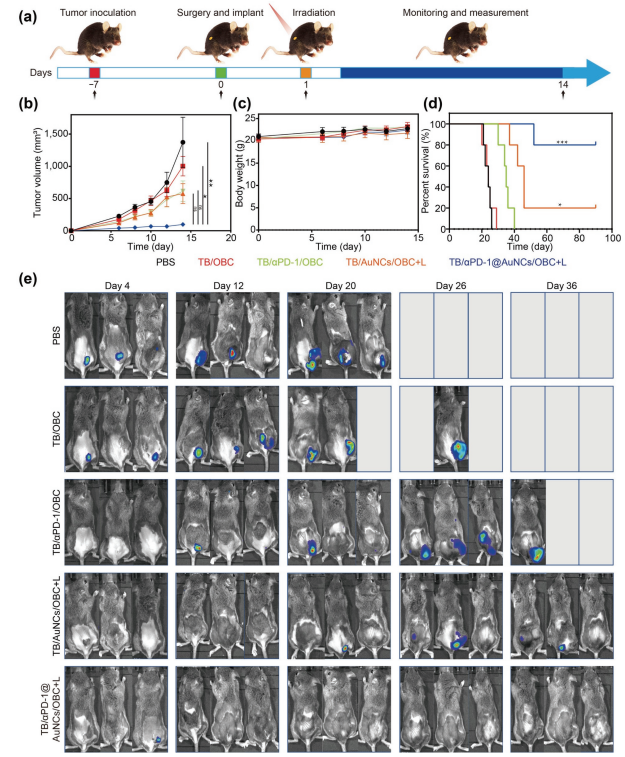
Figure.4. Antitumor efficacy of TB/aPD-1@AuNCs/OBC+L in vivo. (a) The experimental timeline for in vivo treatments.(b) Growth curves of tumor volumes were calculated in different treatment groups (n=5). (c) Body weight variation of SCC7-Luc tumor-bearing mice in different treatment groups. (d) Kaplan-Meier survival curves of SCC7-Luc tumor-bearing mice in different treatment groups.(e) Representative bioluminescence images of SCC7-Luc tumor-bearing mice in different treatment groups at day 4, 12, 20,26, and 36.

Figure.5. TB/aPD-1@AuNCs/OBC+L affects the antitumor immune response by relieving the TME.Lymph nodes and spleen were harvested from SCC7 tumor- bearing mice after various treatments (PBS,TB/OBC,TB/aPD-1/OBC, TB/AuNCs/OBC+L,and TB/aPD-1@AuNCs/OBC+L) for 14 days. Representative flow cytometry zebra plots or histograms (left) and the corresponding quantification analysis (right). (a) Mature CD80'CD86' DCs in lymph nodes gating on CD11c' cells. (b) CD8' T cells gating on CD45'CD3" cells in the spleen.(c) Percentage of MDSCs gating on CD11b' cells in the spleen.(d) IHC staining for CD8* T cells infiltrating the tumor in different treatment groups and the corresponding intensities. (e) IHC staining of the granzyme B released in the tumor in different treatment groups and the corresponding intensities.(f) IHC staining for Ki67 expression in different treatment groups and the corresponding intensities.
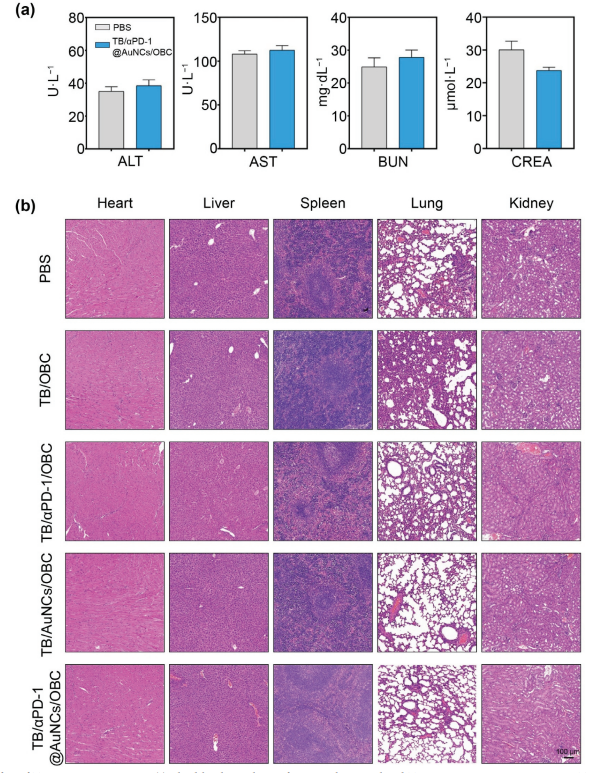
Figure.6. Biosafety of TB/aPD-1@AuNCs/OBC.(a) Blood biochemical test of mice in the control and TB/aPD-1@AuNCs/OBC groups. ALT; AST; BUN; and CREA. Standard deviations were calculated based on data from three mice in each group.(b) H&E staining of the heart, liver, spleen, lung, kidney, and tumor of SCC7tumor-bearing mice after various treatments. Scale bar:100 μm.
该工作由华中科技大学生命科学与技术学院杨光教授团队与武汉大学口腔医院孙志军教授团队共同合作完成。论文共同第一作者为华中科技大学博士生李晓宏、武汉大学博士周俊杰。华中科技大学杨光教授、武汉大学孙志军教授为该文章的共同通讯作者。
该工作得到了国家自然科学基金(批准号 82072996 和 81874131)和中央高校基本科研业务费专项资金(批准号:2042021kf0216、2018M630883、2019T120688)等的支持。
论文链接:https://link.springer.com/article/10.1007/s12274-022-4811-7
论文引用:Zhou, JJ., Li, XH., He, PY. et al. Implantable versatile oxidized bacterial cellulose membrane for postoperative HNSCC treatment via photothermal-boosted immunotherapy. Nano Res. (2022). https://doi.org/10.1007/s12274-022-4811-7.










